News Express: UM research offers novel strategy for immunotherapy of ‘cold’ tumours
新聞快訊:澳大研究為“冷腫瘤”免疫治療提供新策略
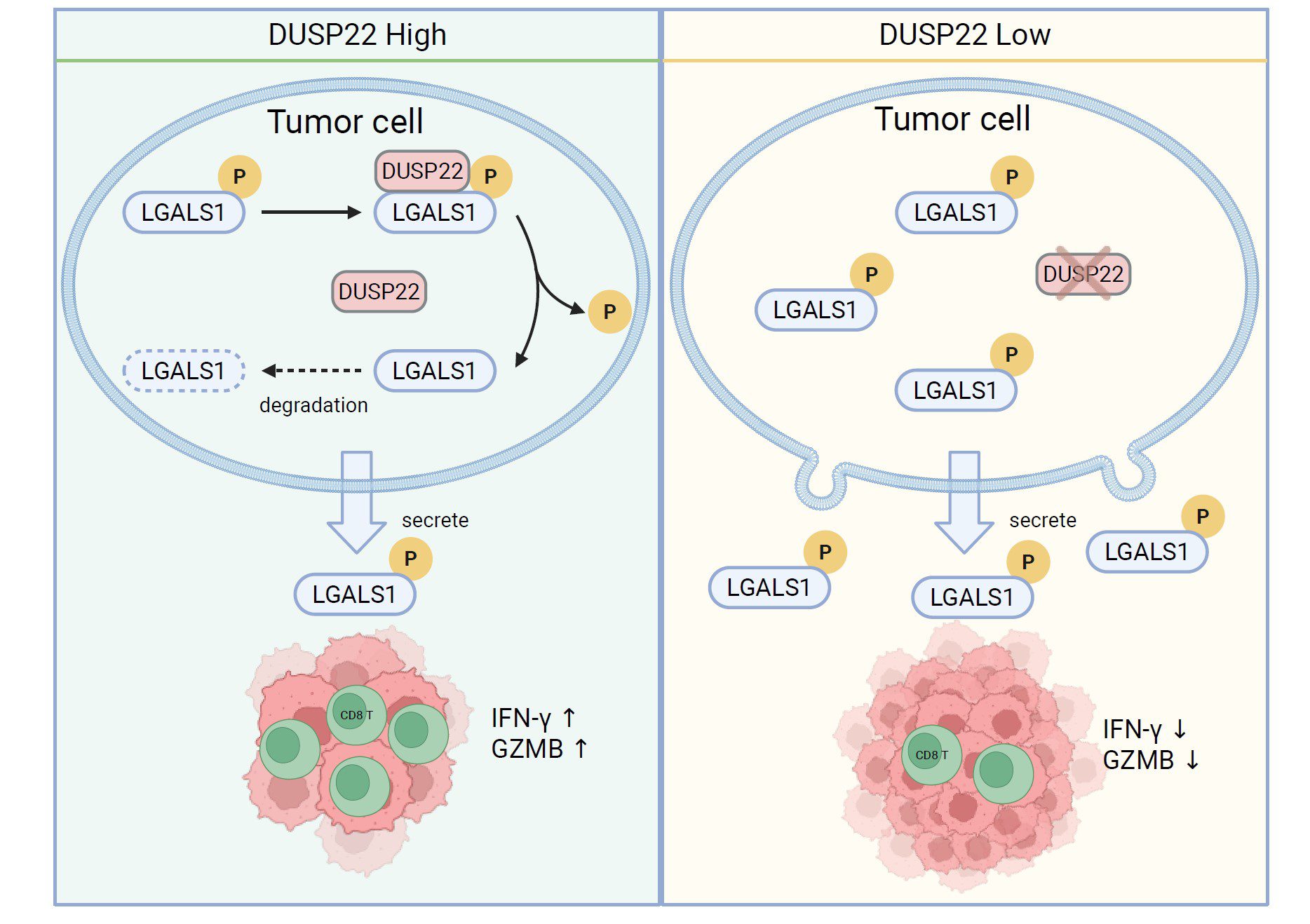
DUSP22-LGALS1軸在腫瘤免疫調節中的示意圖
Schematic illustration of the DUSP22-LGALS1 axis in tumour immune modulation
澳大研究為“冷腫瘤”免疫治療提供新策略
澳門大學健康科學學院講座教授鄧初夏及助理教授苗凱帶領的研究團隊,在腫瘤免疫治療領域取得重要突破。團隊首次揭示了關鍵基因雙特異性磷酸酶22(DUSP22)如何通過“修剪”免疫抑制因子LGALS1的“穩定標籤”,顯著增強T細胞對腫瘤的浸潤能力。這一發現不僅為理解腫瘤免疫逃逸提供了全新視角,也為克服三陰性乳腺癌等“冷腫瘤”的免疫治療耐藥性提出了極具潛力的聯合治療新策略。相關成果已發表於癌症免疫治療協會(Society for Immunotherapy of Cancer)官方期刊《Journal for ImmunoTherapy of Cancer》。
在腫瘤微環境中,CD8⁺ T細胞(一種主要的抗癌免疫細胞)數量不足,是導致免疫治療對許多實體瘤效果不佳的根源。如何將這類免疫細胞稀少的“冷腫瘤”轉化為對治療敏感的“熱腫瘤”,是當前腫瘤免疫治療的核心挑戰。為解決這一難題,團隊運用全基因組篩選技術,在小鼠乳腺癌模型中識別出39個與T細胞浸潤相關的候選基因,當中DUSP22表現最為顯著。實驗證實,DUSP22在乳腺癌組織中的表達水平與CD8⁺ T細胞的浸潤量呈正相關,與腫瘤惡性進展呈負相關。更為關鍵的是,在腫瘤細胞中上調表達DUSP22,能在免疫系統健全的小鼠中顯著抑制腫瘤生長並延長其生存期,而在免疫缺陷小鼠中則無此效果。這如同找到了一個由腫瘤細胞內源性發出、能夠調節免疫細胞對實體腫瘤浸潤的“信號開關”。機制上,團隊首次闡明DUSP22能特異性地去除免疫抑制分子LGALS1上兩個特定點位(Ser8/Thr58)的磷酸基團,使得LGALS1迅速降解。由於LGALS1原本會誘使T細胞凋亡並阻礙其穿越血管壁進入腫瘤內部,它的減少意味著免疫抑制被解除,T細胞得以長驅直入,高效殺滅癌細胞。
基於這一機制,團隊進一步探索了其臨床應用價值,發現使用LGALS1抑制劑(如OTX008)或中和抗體,不僅本身能抑制腫瘤生長,更能與臨床廣泛使用的抗PD-1免疫療法產生顯著協同效應。這種聯合療法成功地將對免疫治療不敏感的“冷腫瘤”轉化為“熱腫瘤”,為破解免疫治療耐藥性提供了新思路。該研究不僅將DUSP22的功能從傳統的信號傳導調控,拓展至腫瘤與免疫系統的“對話”領域,更揭示了LGALS1磷酸化修飾在腫瘤免疫逃逸中的關鍵作用。
該研究通訊作者為鄧初夏與苗凱,第一作者為澳大健康科學學院博士生汪禮建。團隊成員還包括澳門大學、香港大學深圳醫院、澳門科技大學、鄭州大學第三附屬醫院的學者。該項目由澳門特別行政區科學技術發展基金(檔案編號:0073/2021/A2、111/2017/A、0007/2021/AKP、0087/2024/RIB2、0009/2022/AKP、0065/2021/A、0193/2024/AGJ)、澳門大學(檔案編號:MYRG-GRG2023-00150-FHS-UMDF、MYRG-GRG2024-00146-FHS)及國家自然科學基金(檔案編號:82030094)資助。全文可瀏覽:https://doi.org/10.1136/jitc-2025-013142。
欲瀏覽官網版可登入以下連結:
https://www.um.edu.mo/zh-hant/news-and-press-releases/campus-news/detail/63656/
UM research offers novel strategy for immunotherapy of ‘cold’ tumours
A research team led by Chair Professor Chuxia Deng and Assistant Professor Miao Kai in the Faculty of Health Sciences (FHS) at the University of Macau (UM) has made a significant breakthrough in the field of cancer immunotherapy. The study unveils for the first time how the key gene, dual-specificity phosphatase 22 (DUSP22), enhances T cell infiltration into tumours by ‘trimming the stability tag’ of the immunosuppressive factor LGALS1. This discovery not only provides a novel perspective on tumour immune evasion but also proposes a promising combination strategy to overcome immunotherapy resistance in ‘cold’ tumours, such as triple-negative breast cancer (TNBC). The findings have been published in the Journal for ImmunoTherapy of Cancer, the official journal of the Society for Immunotherapy of Cancer (SITC).
In the tumour microenvironment, the scarcity of CD8⁺ T cells—a primary type of anti-cancer immune cell—is a fundamental reason for the poor efficacy of immunotherapies against many solid tumours. Transforming these immune-desert ‘cold’ tumours into treatment-sensitive ‘hot’ tumours is a key challenge in cancer immunotherapy today. To address this, the team used genome-wide screening techniques to identify 39 candidate genes associated with T cell infiltration in a mouse model of breast cancer. Among these, DUSP22 emerged as the most prominent gene. Experiments confirmed that the level of DUSP22 in breast cancer tissues positively correlates with CD8⁺ T cell infiltration and negatively correlates with malignant tumour progression. Crucially, overexpressing DUSP22 in tumour cells significantly suppressed tumour growth and prolonged the survival of mice with a competent immune system, an effect that was completely absent in immunodeficient mice. This is akin to discovering a ‘switch’ within tumour cells that regulates immune cell infiltration into solid tumours. The team also elucidated the mechanism by which DUSP22 specifically removes phosphate groups from two key sites (Ser8/Thr58) on the immunosuppressive molecule LGALS1. This dephosphorylation destabilises LGALS1, leading to its rapid degradation. As LGALS1 typically induces T cell apoptosis and hinders their ability to cross blood vessels and enter tumours, its depletion relieves immunosuppression, allowing T cells to infiltrate tumours unimpeded and kill cancer cells efficiently.
Based on this mechanism, the team further explored its clinical translational potential. They found that LGALS1 inhibitors (such as OTX008) and neutralising antibodies not only inhibit tumour growth independently, but also demonstrate a significant synergistic effect when combined with the widely used anti-PD-1 immunotherapy. This combination therapy successfully transformed immunotherapy-resistant ‘cold’ tumours into responsive ‘hot’ tumours, offering a new avenue to overcome immunotherapy resistance. This research expands the function of DUSP22 beyond traditional signal transduction regulation, establishing its crucial role in the ‘crosstalk’ between tumours and the immune system. It also highlights the critical role of LGALS1 phosphorylation in tumour immune evasion.
The corresponding authors of this study are Prof Deng and Prof Miao. The first author is Wang Lijian, a PhD student in UM FHS. The research team also includes scholars from UM, the University of Hong Kong–Shenzhen Hospital, the Macau University of Science and Technology, and the Third Affiliated Hospital of Zhengzhou University. The project was supported by the Science and Technology Development Fund of the Macao SAR (File Nos.: 0073/2021/A2, 111/2017/A, 0007/2021/AKP, 0087/2024/RIB2, 0009/2022/AKP, 0065/2021/A, 0193/2024/AGJ), the University of Macau (File Nos.: MYRG-GRG2023-00150-FHS-UMDF, MYRG-GRG2024-00146-FHS), and the National Natural Science Foundation of China (File No.: 82030094). The full article is available at: https://doi.org/10.1136/jitc-2025-013142
To read the news on UM’s official website, please visit the following link:
https://www.um.edu.mo/news-and-press-releases/campus-news/detail/63656/