News Express: UM research team advances personalised osteoporosis screening
新聞快訊:澳大團隊推動精準化骨質疏鬆個性化篩查
研究團隊將多基因風險評分(PRS)納入篩查決策體系
The research team incorporates polygenic risk scores (PRS) into the screening decision-making system
澳大團隊推動精準化骨質疏鬆個性化篩查
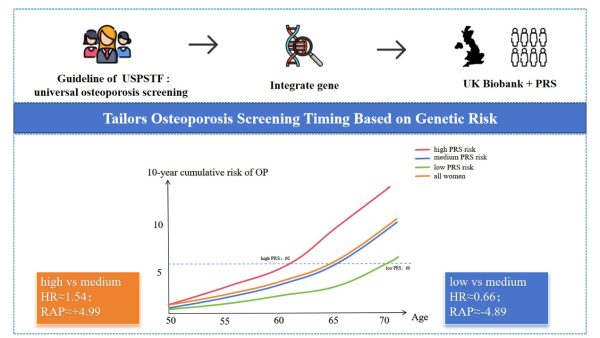
澳門大學健康科學學院教授紀建光的研究團隊在骨質疏鬆個性化篩查研究取得重要進展。該研究創新性地將多基因風險評分(PRS)納入篩查決策體系,依據個體遺傳背景實現風險分層與精準預防。這一策略突破了當前主要依賴固定年齡閾值、忽視遺傳差異的局限,從“一刀切”到“因人而異”,既提升了中老年女性骨折風險的早期識別能力,也減少了不必要的篩查與資源浪費,相關成果已於知名期刊《BMC Medicine》發表。
隨著全球人口老齡化加速,骨質疏鬆及相關骨折已成為重大公共衛生挑戰。據統計,全球每年發生約890萬例骨質疏鬆性骨折,其中髖部骨折在一年內的死亡率高達20%至24%。大規模全基因組關聯研究(GWAS)顯示,遺傳因素在骨質疏鬆發生中起到關鍵作用,目前已發現數百個與骨密度及骨折風險相關的遺傳位點。然而,現行篩查方案仍普遍沿用“一刀切”模式——通常建議所有65歲以上女性接受篩查,這可能導致高遺傳風險人群被“延遲診斷”,而低風險人群則面臨“過度篩查”,造成醫療資源配置不均。如何實現骨質疏鬆的個體化、精準化篩查,已成為公共衛生領域亟待解決的問題。
多基因風險評分(PRS)通過整合大量常見遺傳變異的微小效應,可對個體遺傳易感性進行量化評估,已在不同疾病領域展現出轉化潛力。該研究基於UK Biobank資料,利用PRS量化個體遺傳風險,將統一的篩查年齡轉化為個性化決策節點。結果顯示,與中等遺傳風險女性相比,高遺傳風險女性骨質疏鬆發生時間平均提前5年,建議將篩查年齡提前至60歲;而低遺傳風險女性則可延後約4年進行篩查,即延遲至69歲。對高風險人群前移篩查,有助於在臨床前期識別更多患者,降低後續骨折及相關殘疾、死亡風險與經濟負擔;對低風險人群適度延後篩查,則可提升整體篩查的效率與成本效益。佇列模型估算表明,對高遺傳風險女性提前篩查可預防骨折總數的41%,結合遺傳風險的篩查策略亦顯著提高了篩查效率。在現有策略中,平均每篩查28人可發現1例骨折,而在高遺傳風險組中,僅需篩查22人即可發現1例。
該研究首次將遺傳信息系統融入骨質疏鬆早期篩查的時間決策中,把基於人群平均風險的篩查閾值轉化為個體化的干預節點,為推動骨質疏鬆防治從普遍篩查邁向精準分層提供了紥實證據。這一方向不僅契合衰老相關疾病的異質性特點,有助於實現早期干預與前瞻性健康管理;同時通過精準聚焦高危人群、合理調整低危人群篩查時機,有望在優化醫療資源配置的同時,更有效地緩解老齡化社會相關疾病負擔,具有重要的學術價值與實踐意義。
該研究通訊作者為紀建光,其博士生王冬雪為第一作者,澳大健康科學學院研究助理孫雯為共同第一作者;北京大學人民醫院護理部副主任李曉丹、隆德大學初級衛生保健研究中心副教授王曉為共同通訊作者。澳大健康科學學院博士後劉迪、福建醫科大學附屬婦產醫院副教授衣歡也對研究做出了重要貢獻。該項目由澳門大學發展基金會和澳門大學(檔案編號:UMDF-TISF/2025/001/FHS和FHS-CC-046-001-2024)資助。全文可瀏覽:https://doi.org/10.1186/s12916-025-04601-1。
欲瀏覽官網版可登入以下連結:
https://www.um.edu.mo/zh-hant/news-and-press-releases/campus-news/detail/63519/
UM research team advances personalised osteoporosis screening
A research team led by Ji Jianguang, professor in the Faculty of Health Sciences (FHS) at the University of Macau (UM), has made significant progress in the study of personalised osteoporosis screening. The study incorporates a polygenic risk score (PRS) into the screening decision-making system for the first time, enabling risk stratification and precision prevention based on individual genetic backgrounds. This strategy overcomes the limitations of existing guidelines, which primarily rely on fixed age thresholds and overlook genetic differences. It shifts the focus from a ‘one-size-fits-all’ approach to a ‘personalised’ model of care. It not only enhances the early identification of fracture risk in middle-aged and elderly women but also reduces unnecessary screening and wastage of resources. The research findings have been published in the renowned journal BMC Medicine.
Against the backdrop of an ageing global population, osteoporosis and related fractures have become a major public health concern. Statistics show that approximately 8.9 million osteoporotic fractures occur worldwide each year, and hip fractures carry a mortality rate of 20–24% within one year of injury. Large-scale Genome-Wide Association Studies (GWAS) indicate that genetic factors play a crucial role in the onset of osteoporosis, and hundreds of genetic loci associated with bone mineral density and fracture risk have already been identified. However, current screening protocols still predominantly follow a ‘one-size-fits-all’ approach, typically recommending screening for all women over 65. This approach may result in ‘delayed diagnoses’ for individuals with high genetic risk and ‘over-screening’ for those with low risk, leading to the inefficient allocation of medical resources. Therefore, achieving individualised and precise osteoporosis screening has become an urgent issue in public health.
A polygenic risk score (PRS) quantifies an individual’s genetic susceptibility by aggregating the small effects of numerous common genetic variants, and has shown translational potential in various disease areas. This study, based on data obtained from the UK Biobank, used PRS to quantify individual genetic risk, transforming a uniform screening age into personalised medical decisions. The results showed that, compared to women with average genetic risk, those with high genetic risk developed osteoporosis on average five years earlier. This suggests that screening should begin at the age of 60. Conversely, women with low genetic risk could postpone screening by approximately four years, to age 69. Earlier screening of high-risk individuals will help identify more patients in the pre-clinical stage, thereby reducing the risk of subsequent fractures, as well as the associated disability, mortality, and economic burden. Delaying screening appropriately for low-risk individuals will improve the overall efficiency and cost-effectiveness of screening programmes. Early screening of high-genetic-risk women could prevent 41% of total fractures, and incorporating genetic risk information into screening programmes could significantly improve their efficiency. The results demonstrated that the number needed to screen (NNS) to prevent one fracture was 22 in the high-risk group, making it more efficient than an NNS of 28 in the overall population.
This study is the first to systematically integrate genetic information into decisions about the timing of early osteoporosis screening, and transform screening thresholds based on population-average risk into personalised interventions. It provides robust evidence to support shifting osteoporosis prevention from universal screening to precise risk stratification, facilitating early intervention and proactive health management. By focusing on high-risk individuals and adjusting the screening age for low-risk populations, it is hoped to optimise the allocation of healthcare resources and alleviate the disease burden associated with an ageing society more effectively, demonstrating significant academic and practical implications.
The study’s corresponding author is Prof Ji, with his PhD student Wang Dongxue as first author, and Sun Wen, a research assistant in UM FHS, as co-first author. Li Xiaodan, deputy director of the Department of Nursing at Peking University People’s Hospital, and Wang Xiao, associate professor in the Center for Primary Health Care Research at Lund University, are co-corresponding authors. Liu Di, a postdoctoral fellow in UM FHS, and Yi Huan, associate professor at Affiliated Hospital of Fujian Medical University, also made important contributions to the research. The project was supported by the University of Macau (File Nos.: UMDF-TISF/2025/001/FHS and FHS-CC-046-001-2024). The full version of the research article is available at: https://doi.org/10.1186/s12916-025-04601-1.
To read the news on UM’s official website, please visit the following link:
https://www.um.edu.mo/news-and-press-releases/campus-news/detail/63519/