News Express: UM researchers reveal key to breast cancer’s resistance to targeted drugs
新聞快訊:澳大破解乳腺癌靶向藥耐藥關鍵
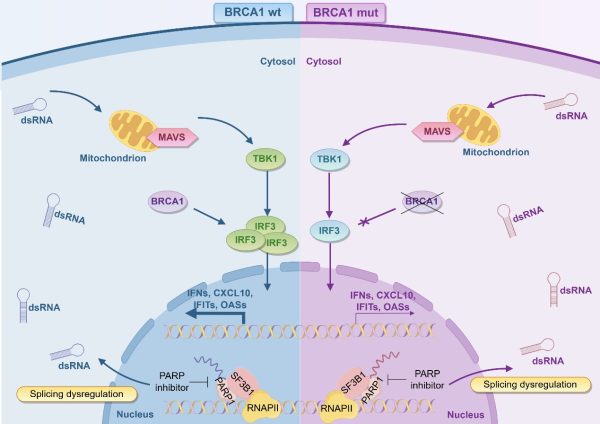
BRCA1缺陷通過抑制IRF3介導的免疫信號通路導致PARP抑制劑耐藥
BRCA1 deficiency downregulates IRF3-mediated innate immune response, leading to resistance to PARP inhibitors
澳大破解乳腺癌靶向藥耐藥關鍵
澳門大學健康科學學院講座教授鄧初夏與教授張仲榮帶領的研究團隊揭示了BRCA1缺陷乳腺癌對靶向藥物PARP抑制劑產生耐藥性的關鍵分子機制,為克服此類抑制劑的耐藥性提供了科學依據,並進一步提出相關治療策略。研究成果已發表於國際知名期刊《蛋白質與細胞》(Protein & Cell)。
乳腺癌是全球女性最常見的惡性腫瘤,其發病與遺傳因素密切相關。約10%的病例具有家族遺傳背景,其中大部分由BRCA1或BRCA2基因突變引起。BRCA1蛋白在DNA同源重組修復中具有關鍵作用,該修復途徑能精準修復雙鏈斷裂,維持基因組穩定。若其功能喪失,細胞將依賴其他容易出錯的修復機制,導致基因組不穩定和癌變。基於這一缺陷,PARP抑制劑被開發為靶向藥物,通過“合成致死”機制選擇性殺傷DNA修復缺陷的癌細胞,現已成為治療BRCA突變乳腺癌的有效手段。然而,臨床耐藥性是制約其療效的主要挑戰。
為應對耐藥,研究揭示了DNA損傷啟動先天免疫反應的新機制。DNA損傷不僅威脅基因組穩定,還會產生胞質DNA片段和異常雙鏈RNA(dsRNA)。前者通過cGAS-STING通路誘導干擾素產生,增強抗腫瘤免疫反應;後者則觸發MAVS介導的類抗病毒先天免疫反應。相關發現開闢了聯合治療新方向——STING激動劑與PARP抑制劑聯用已顯示出協同效應。雖然dsRNA-MAVS通路的具體機制尚待闡明,但將靶向DNA修復與啟動免疫應答相結合,有望為克服耐藥、提升乳腺癌治療效果提供突破性策略。
澳大團隊通過功能蛋白質組學發現,PARP抑制劑可增強PARP1與剪接體因數SF3B1的相互作用,干擾正常剪接體功能,導致dsRNA的積累並啟動類抗病毒先天免疫反應。研究同時揭示BRCA1具有調控先天免疫的新功能,其缺失會導致IRF3表達下調,削弱該免疫應答,從而降低細胞對PARP抑制劑的敏感性,形成固有耐藥機制。值得注意的是,dsRNA類似物poly(I:C)可有效啟動該免疫通路,與PARP抑制劑聯用能顯著增強對BRCA1缺陷腫瘤細胞的殺傷效果。動物實驗證實,該聯合方案可顯著提高腫瘤抑制率。該研究闡明了BRCA1缺陷通過抑制IRF3介導的免疫信號通路導致耐藥的新機制,並創新性地提出通過poly(I:C)啟動該通路以逆轉耐藥,為臨床克服PARP抑制劑耐藥提供了新策略。
該研究通訊作者為鄧初夏及張仲榮,第一作者為該學院博士生張翠婷,其他團隊成員包括學院副教授潘全威、客席副教授徐曉玲、助理教授苗凱、博士後周靖波及雷海鵬。該研究獲國家自然科學基金(檔案編號:82030094)、澳門特別行政區科學技術發展基金(檔案編號:0009/2022/AKP、0054/2023/RIA1、0129/2024/RIA2、0137/2020/A3)和澳門大學(檔案編號:MYRG-GRG2024-00073-FHS)資助。全文可瀏覽:https://academic.oup.com/proteincell/advance-article/doi/10.1093/procel/pwaf104/8419885?login=false。
欲瀏覽官網版可登入以下連結:
https://www.um.edu.mo/zh-hant/news-and-press-releases/campus-news/detail/63566/
UM researchers reveal key to breast cancer’s resistance to targeted drugs
A research team led by Chair Professor Chuxia Deng and Professor Edwin Cheung in the Faculty of Health Sciences (FHS) at the University of Macau (UM) has revealed the key molecular mechanism behind resistance to the targeted drug PARP inhibitors in BRCA1-deficient breast cancer, providing a scientific basis for overcoming this type of drug resistance. The team also proposed a related therapeutic strategy to address this resistance. The research has been published in the internationally renowned journal Protein & Cell.
Breast cancer is the most common malignant tumour among women worldwide, and its development is closely linked to genetic factors. About 10% of cases have a familial genetic background, most of which are caused by mutations in the BRCA1 or BRCA2 genes. BRCA1 plays a key role in the process of DNA homologous recombination repair, a repair pathway which is responsible for accurately repairing double-strand breaks and maintaining genomic stability. If this function is impaired, cells rely on other error-prone repair mechanisms, which can lead to genomic instability and cancer development. Based on this defect, PARP inhibitors have been developed as targeted drugs that selectively kill cancer cells with DNA repair defects through a ‘synthetic lethality’ mechanism. They have now become an effective therapy for treating BRCA-mutated breast cancer. However, clinical resistance remains a major challenge that limits their efficacy.
To overcome drug resistance, the research team has revealed a new mechanism through which DNA damage activates innate immune responses. DNA damage not only threatens genomic stability, but also generates cytosolic DNA fragments and abnormal double-stranded RNA (dsRNA). DNA fragments can induce interferon production through the cGAS-STING pathway, thereby enhancing anti-tumour immune responses. Meanwhile, dsRNA triggers MAVS-mediated anti-viral mimicry innate immune responses. These findings open new avenues for combination therapy involving the use of STING agonists alongside PARP inhibitors. Although the detailed mechanisms of the dsRNA-MAVS pathway are yet to be elucidated, the combination of targeted DNA repair and the activation of immune responses shows promise in overcoming resistance and improving breast cancer treatment outcomes.
Through functional proteomics, the research team discovered that PARP inhibitors can enhance the interaction between PARP1 and the splicing factor SF3B1. This disrupts spliceosome function, leading to the accumulation of dsRNA and the activation of an anti-viral mimicry innate immune response. The study also revealed a new function of BRCA1 in regulating innate immunity. BRCA1 deficiency leads to the downregulation of IRF3 expression, thereby weakening this immune response and reducing the sensitivity to PARP inhibitors, resulting in intrinsic resistance. Notably, the dsRNA analogue poly(I:C) can effectively activate this immune pathway, and combining it with PARP inhibitors significantly enhances their killing effect on BRCA1-deficient breast cancer cells. Animal experiments confirmed that this combination strategy can significantly improve the tumour suppression rate. This study elucidates a new mechanism by which BRCA1 deficiency leads to resistance by downregulating the IRF3-mediated innate immune signalling pathway. It also puts forward the innovative approach of reversing this resistance by activating the pathway with poly(I:C), providing a new strategy for overcoming PARP inhibitor resistance in BRCA1-deficient breast cancer.
The corresponding authors of this study are Prof Deng and Prof Cheung, with Zhang Cuiting, PhD candidate in UM FHS, as the first author. Other team members include Associate Professor Terence Poon Chuen Wai, Adjunct Associate Professor Xu Xiaoling, Assistant Professor Miao Kai, and postdoctoral fellows Zhou Jingbo and Josh Lei Haipeng in UM FHS. The study was supported by the Natural Science Foundation of China (File No.: 82030094), the Science and Technology Development Fund of the Macao SAR (File Nos.: 0009/2022/AKP, 0054/2023/RIA1, 0129/2024/RIA2, and 0137/2020/A3), and the University of Macau (File No.: MYRG-GRG2024-00073-FHS). The full version of the research article is available at: https://academic.oup.com/proteincell/advance-article/doi/10.1093/procel/pwaf104/8419885?login=false.
To read the news on UM’s official website, please visit the following link:
https://www.um.edu.mo/news-and-press-releases/campus-news/detail/63566/