News Express: UM research team identifies novel precision drug target for VHL-deficient kidney cancer
新聞快訊:澳大團隊發現VHL缺失型腎癌新型精準藥物靶點
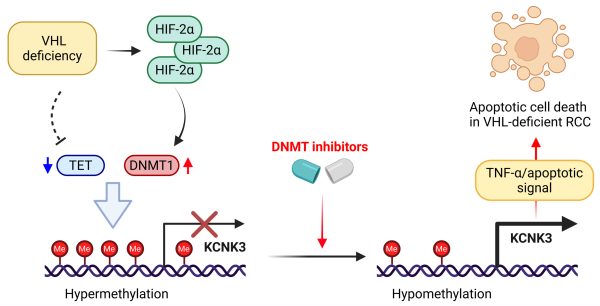
VHL缺失腎癌對DNMT抑制劑呈現選擇性脆弱性的推測機制
Proposed mechanism underlying the selective vulnerability of VHL-deficient kidney cancer to DNMT inhibitors
澳大團隊發現VHL缺失型腎癌新型精準藥物靶點
澳門大學健康科學學院副教授沈仲燮帶領研究團隊,聯同重慶醫科大學、上海交通大學醫學院附屬仁濟醫院發現VHL缺失型腎細胞癌(RCC)的全新治療弱點。研究顯示,缺乏腫瘤抑制基因VHL的RCC細胞對DNA甲基轉移酶(DNMT)抑制劑極為敏感,為腎癌亞型提供一項具潛力的精準醫療策略。相關成果已發表於國際期刊《Experimental & Molecular Medicine》。
腎細胞癌常由VHL基因缺失驅動,導致HIF‑2α穩定並重塑細胞代謝與血管生成。近年研究指出,VHL失活亦會引發廣泛的DNA高甲基化。為利用這種表觀遺傳狀態進行治療,研究團隊利用VHL同基因背景的RCC模型進行合成致死藥物篩選。結果顯示地西他濱(Decitabine)、阿扎胞苷(Azacitidine)等已獲FDA批准的DNMT抑制劑,以及RX‑3117、SGI‑1027等研發中藥物,均能選擇性抑制VHL缺失型RCC細胞的生長。
機制研究揭示,VHL缺失會透過HIF‑2α上調DNMT1,導致廣泛的CpG高甲基化。在被該表觀遺傳重塑所沉默的基因中,研究團隊鑑定出鉀離子通道腫瘤抑制基因KCNK3是DNMT抑制劑誘導合成致死的關鍵媒介。KCNK3的啟動子在VHL缺失型RCC中高度甲基化並被抑制,而DNMT抑制劑可逆轉此甲基化,恢復KCNK3表達並啟動細胞凋亡訊號。
在小鼠異種移植腫瘤模型中,地西他濱能選擇性抑制VHL缺失型RCC腫瘤的生長;然而當KCNK3被敲低時,抑制效果大幅減弱,顯示出高度的治療特異性。研究團隊亦在患者來源的RCC細胞模型中驗證此發現:帶有致病性VHL突變的腫瘤細胞對DNMT抑制劑的敏感度明顯高於VHL野生型樣本。
研究提供了有力證據,表明DNMT抑制是針對VHL缺失型RCC的一項合成致死策略,為帶有此常見基因變異的患者開啟新的治療方向。透過整合合成致死篩選、表觀遺傳分析、機制研究及患者來源模型,是次研究為未來臨床轉化奠定了堅實基礎。
該研究通訊作者為沈仲燮、重慶醫科大學藥學院院長黨永軍及上海交通大學醫學院附屬仁濟醫院主任醫師王建峰,第一作者為澳大健康科學學院博士生蒲悅及重慶醫科大學王子若宇,澳大健康科學學院副教授李剛、助理教授苗凱、高級導師陳凱靈等為研究作出貢獻。該研究獲澳門特別行政區科學技術發展基金(檔案編號:0139/2024/AFJ、0049/2022/A)、深圳市科技創新委員會(檔案編號:EF2023‑00070‑FHS)、澳門大學發展基金會和澳門大學(檔案編號:MYRG‑GRG2023‑00124‑FHS‑UMDF、MYRG‑GRG2024‑00056‑FHS、MYRG‑GRG2025‑00182‑FHS)、澳門特別行政區科學技術發展基金與國家自然科學基金委員會聯合科研資助計劃(檔案編號:22461160267)及國家教育部澳門大學精準腫瘤學前沿科學中心(檔案編號:SP2023-00001-FSCPO)資助。全文可瀏覽:https://www.nature.com/articles/s12276-026-01663-w。
欲瀏覽官網版可登入以下連結:
https://www.um.edu.mo/zh-hant/news-and-press-releases/campus-news/detail/63946/
UM research team identifies novel precision drug target for VHL-deficient kidney cancer
A research team led by Shim Joong Sup, associate professor in the Faculty of Health Sciences (FHS) at the University of Macau (UM), in collaboration with researchers from Chongqing Medical University and Renji Hospital of Shanghai Jiaotong University, has discovered an unrecognised therapeutic vulnerability in VHL‑deficient renal cell carcinoma (RCC), the most common form of kidney cancer. The study shows that RCC cells lacking the tumour suppressor von Hippel-Lindau (VHL) are highly sensitive to DNA methyltransferase (DNMT) inhibitors, revealing a promising precision medicine strategy for the major RCC subtype. The findings have been published in the international journal Experimental & Molecular Medicine.
Renal cell carcinoma (RCC) is often caused by the loss of the VHL gene, which stabilises HIF‑2α and alters cellular metabolism and angiogenesis. Recent studies have also suggested that VHL inactivation triggers widespread DNA hypermethylation. In order to leverage this epigenetic state to enhance therapeutic efficiency, the research team performed a synthetic lethal drug screen using VHL‑isogenic RCC models. The results showed that FDA‑approved DNMT inhibitors, such as decitabine and azacitidine, as well as the investigational agents RX‑3117 and SGI‑1027, selectively suppress the growth of VHL‑deficient RCC cells.
Mechanistic studies revealed that VHL deficiency results in HIF‑2α-driven upregulation of DNMT1, which leads to widespread CpG hypermethylation. Among the genes silenced by this epigenetic remodelling, the team identified KCNK3, a potassium‑channel tumour suppressor, as a key mediator of synthetic lethality induced by DNMT inhibitors. The KCNK3 promoter is hypermethylated and repressed in VHL‑deficient RCC, but DNMT inhibitors can reverse this methylation, thereby restoring KCNK3 expression and triggering apoptotic signalling.
In mouse xenograft models, decitabine was found to selectively inhibit the growth of VHL‑deficient RCC tumours. However, this effect was largely abolished when KCNK3 was depleted, demonstrating strong therapeutic specificity. The team further validated this finding using patient‑derived RCC cell models, in which tumour cells harbouring pathogenic VHL mutations exhibited markedly higher sensitivity to DNMT inhibitors than VHL‑wildtype samples.
This study provides compelling evidence that DNMT inhibition represents a synthetic lethal strategy for VHL‑deficient RCC, offering a new therapeutic avenue for patients with this common genetic alteration. Through the integration of synthetic lethal screening, epigenetic profiling, mechanistic dissection, and patient‑derived models, the research establishes a strong foundation for future clinical translation.
The corresponding authors of the study are Prof Shim, Dang Yongjun, dean of the School of Pharmacy at Chongqing Medical University, and Wang Jianfeng, chief physician at Renji Hospital of Shanghai Jiao Tong University. The first authors are Pu Yue, a PhD candidate at UM, and Wang Ziruoyu from Chongqing Medical University. Other contributors to the research include Associate Professor Li Gang, Assistant Professor Miao Kai, and Senior Instructor Tan Kaeling in UM FHS. This project was funded by the Science and Technology Development Fund of the Macao SAR (Grant Nos.: 0139/2024/AFJ and 0049/2022/A), the Shenzhen Science and Technology Innovation Commission (Grant No.: EF2023‑00070‑FHS), the University of Macau Development Foundation and the University of Macau (Grant Nos.: MYRG‑GRG2023‑00124‑FHS‑UMDF, MYRG‑GRG2024‑00056‑FHS, and MYRG‑GRG2025‑00182‑FHS), Joint Funding Scheme for NSFC‑FDCT Project (Grant No.: 22461160267), and the Ministry of Education Frontiers Science Center for Precision Oncology, University of Macau (Grant No.: SP2023-00001-FSCPO). The full version of the research article is available at: https://www.nature.com/articles/s12276-026-01663-w.
To read the news on UM’s official website, please visit the following link:
https://www.um.edu.mo/news-and-press-releases/campus-news/detail/63946/